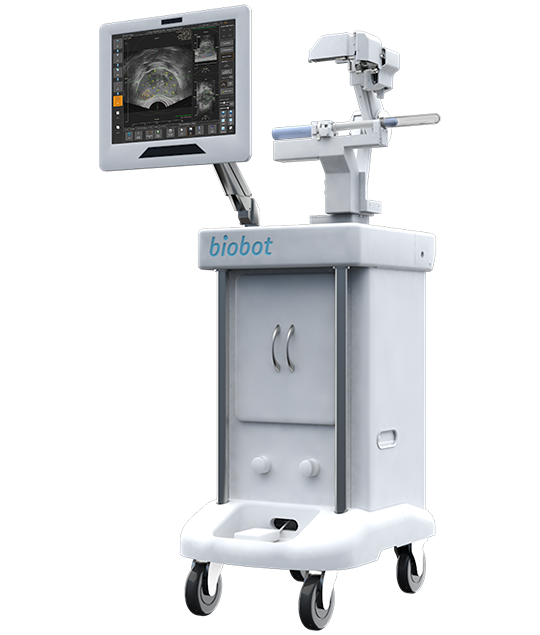
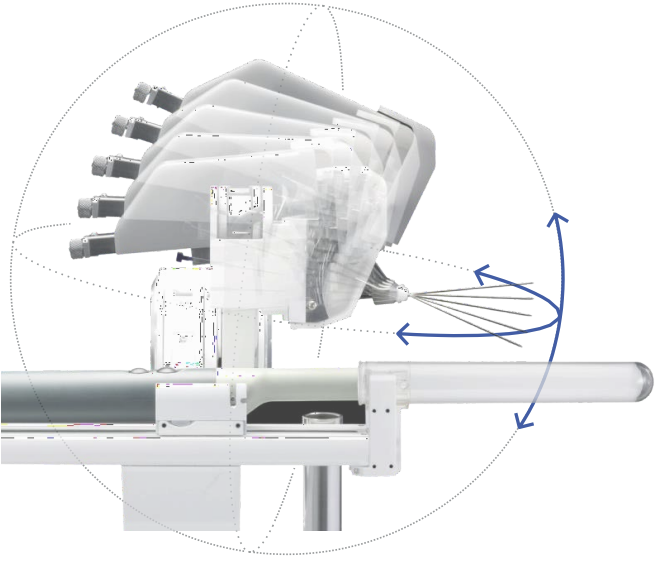
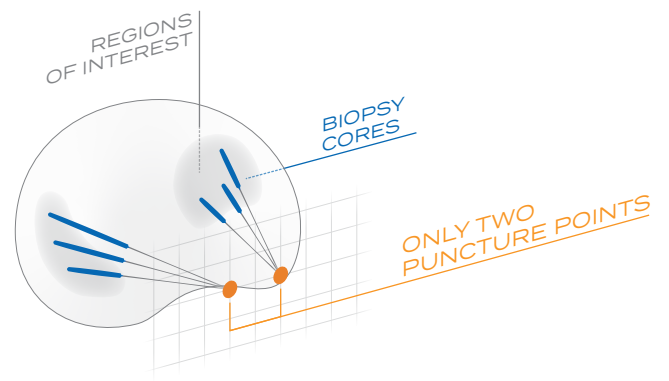
iSR’OBOT™ MONA LISA
This device fuses MRI with real-time ultrasound to generate 3D visualizations, enalbling precise targeting of suspected lesions during prostate biopsy and improving the detection rate of prostate cancer.
- U.S. Food and Drug Administration (FDA) Approved in 2013
- The Ministry of Food and Drug Safety (MFDS) Approved in August 2025